3 results
Pharmacovigilance analysis of the Vigibase on antidepressants-related withdrawal syndrome in adults and adolescents
- C. Gastaldon, G. Schoretsanitis, E. Arzenton, E. Raschi, D. Papola, G. Ostuzzi, U. Moretti, E. Seifritz, J. M. Kane, G. Trifirò, C. Barbui
-
- Journal:
- European Psychiatry / Volume 66 / Issue S1 / March 2023
- Published online by Cambridge University Press:
- 19 July 2023, pp. S95-S96
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Introduction
Antidepressant discontinuation may cause withdrawal syndrome in some cases. However, evidence on this syndrome related to individual antidepressants is limited, as well as about individual risk factors for severe reactions.
ObjectivesTo ascertain whether each individual antidepressant is associated with an increased reporting of withdrawal syndrome as compared with other medications, and to examine clinical risk factors for severe reactions.
MethodsWe conducted a pharmacovigilance study, with a case/non-case design. We included reports of antidepressant-related withdrawal syndrome from the VigiBase, the WHO global database of individual case safety reports of suspected adverse drug reactions. We performed a disproportionality analysis (calculating reporting odds ratio (ROR) and the Bayesian information component (IC)) of reports of antidepressant-related withdrawal syndrome, comparing antidepressants to all other drugs and to buprenorphine (as a positive control). Antidepressants with significant disproportionate reporting were ranked in terms of clinical priority. We compared serious versus non-serious reactions to determine clinical risk factors for severe reactions.
ResultsBased on 31,688 reports of antidepressant-related withdrawal syndrome, we detected a disproportionate reporting for 23 antidepressants. The ROR for antidepressants altogether, compared to all other drugs, was 14.26 (95%CI:14.08-14.45), 17.01 for other antidepressants (95%CI:16.73-17.29), 13.65 for SSRIs (95%CI:13.41-13.90) and 2.8 for tricyclics (95%CI:2.59-3.02). Based on clinical priority ranking, the strongest signals were found for paroxetine, duloxetine, venlafaxine and desvenlafaxine (figure 1), being comparable to buprenorphine. Severe reactions were more frequently reported in males, adolescents, persons with multiple medications, and with longer treatment duration.
Image:
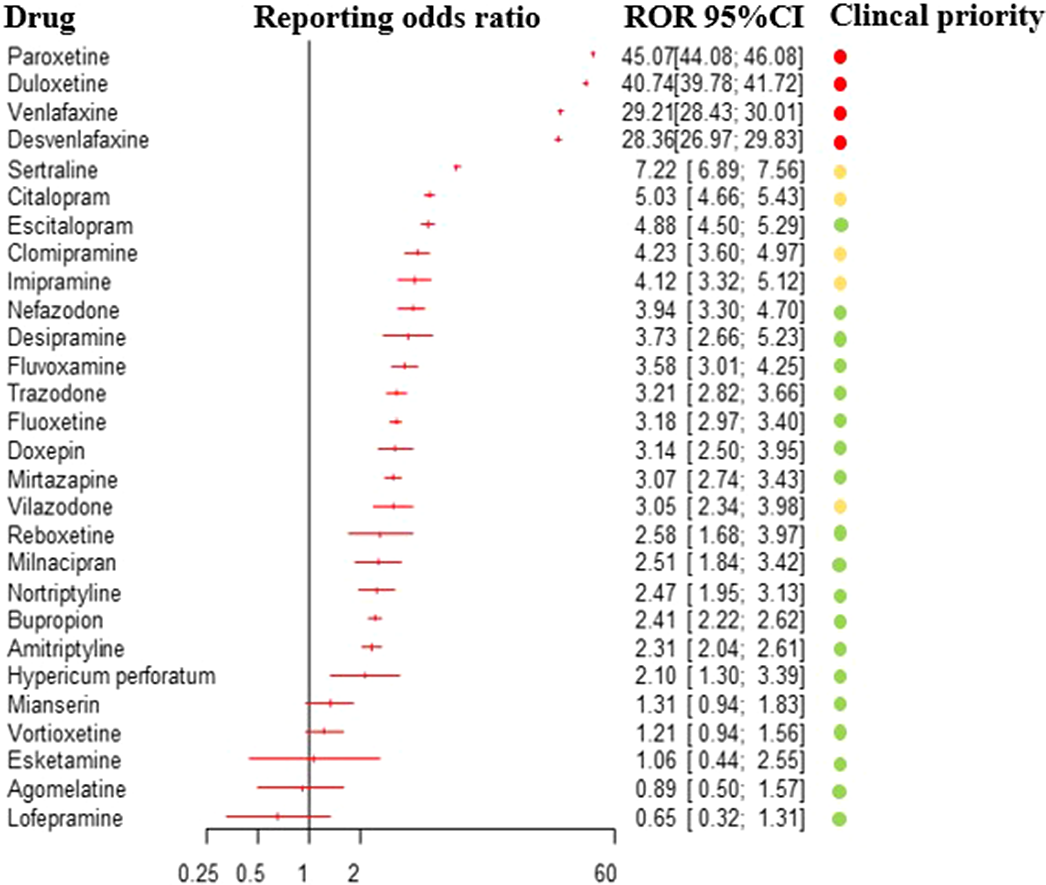 Conclusions
ConclusionsAntidepressants are associated with increased reporting of withdrawal syndrome compared with other medications, with differences between individual antidepressants. Clinicians should be aware of such differences, when prescribing and discontinuing these drugs, as well as of the risk to experience more severe withdrawal symptoms in specific cases.
Disclosure of InterestC. Gastaldon: None Declared, G. Schoretsanitis Consultant of: Dr. Schoretsanitis has served as a consultant for HLS Therapeutics, E. Arzenton : None Declared, E. Raschi : None Declared, D. Papola: None Declared, G. Ostuzzi: None Declared, U. Moretti: None Declared, E. Seifritz Grant / Research support from: Dr. Seifritz has received educational grants, consulting fees and lecture honoraria from Janssen Cilag, Lundbeck, Angelini, Otsuka, Servier, Ricordati, Vifor, Sunovion, Schwabe and Mepha, Consultant of: Dr. Seifritz has received educational grants, consulting fees and lecture honoraria from Janssen Cilag, Lundbeck, Angelini, Otsuka, Servier, Ricordati, Vifor, Sunovion, Schwabe and Mepha, J. Kane Shareolder of: LB Pharmaceuticals and Vanguard Research Group, Consultant of: Dr. Kane has been a consultant and/or advisor for or has received honoraria from Alkermes, Allergan, LB Pharmaceuticals, H. Lundbeck, Intracellular Therapies, Janssen Pharmaceuticals, Johnson and Johnson, Merck, Minerva, Neurocrine, Newron, Otsuka, Pierre Fabre, Reviva, Roche, Sumitomo Dainippon, Sunovion, Takeda, Teva and UpToDate, G. Trifirò Grant / Research support from: he was the scientific director of a Master program on pharmacovigilance, pharmacoepidemiology and real-world evidence which has received non-conditional grant from various pharmaceutical companies; he coordinated a pharmacoepidemiology team at the University of Messina until Oct 2020, which has received funding for conducting observational studies from various pharmaceutical companies (Boehringer Ingelheim, Daichii Sankyo, PTC Pharmaceuticals). He is also scientific coordinator of the academic spin-off “INSPIRE srl” which has received funding for conducting observational studies from contract research organizations (RTI Health Solutions, Pharmo Institute N.V.)., Consultant of: Dr. Trifirò has served in the last three years on advisory boards/seminars funded by SANOFI, Eli Lilly, AstraZeneca, Abbvie, Servier, Mylan, Gilead, Amgen;, Speakers bureau of: Dr. Trifirò has served in the last three years on advisory boards/seminars funded by SANOFI, Eli Lilly, AstraZeneca, Abbvie, Servier, Mylan, Gilead, Amgen;, C. Barbui: None Declared
Pharmacovigilance analysis of the Vigibase on neonatal withdrawal syndrome following in utero exposure to antidepressants
- C. Gastaldon, E. Arzenton, E. Raschi, O. Spigset, D. Papola, G. Ostuzzi, U. Moretti, G. Trifirò, C. Barbui, G. Schoretsanitis
-
- Journal:
- European Psychiatry / Volume 66 / Issue S1 / March 2023
- Published online by Cambridge University Press:
- 19 July 2023, p. S96
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Introduction
Evidence on neonatal withdrawal syndrome following antidepressant intrauterine exposure is limited, particularly for antidepressants other than selective serotonin reuptake inhibitors (SSRIs).
ObjectivesTo ascertain whether maternal antidepressant treatment may be associated with withdrawal syndrome in neonates, investigating the comparative reporting between individual antidepressants and classes.
MethodsWe performed a case/non-case pharmacovigilance study, searching reports of withdrawal syndrome in newborns in the VigiBase, the WHO database of suspected adverse drug reactions. Disproportionality analysis was performed, estimating reporting odds ratio (ROR) and the Bayesian information component (IC). Antidepressants were compared to all other medications, to methadone, and within each class of antidepressants (SSRIs, tricyclics (TCA) and other antidepressants). Antidepressants were ranked in terms of clinical priority, based on a semiquantitative score.
ResultsWe retrieved 406 reports of neonatal withdrawal syndrome in 379 neonates related to 15 antidepressants. Compared to all other drugs, disproportionate reporting was detected for antidepressants altogether (ROR: 6.18, 95%CI:5.45-7.01), for TCAs (10.55, 95%CI:8.02-13.88), other antidepressants (ROR: 5.90, 95%CI:4.74-7.36) and SSRIs (ROR: 4.68, 95%CI:4.04-5.42). All antidepressants showed a significant disproportionality, apart from bupropion (figure 1). We did not find any disproportionate reporting for any antidepressant compared to methadone. The clinical priority ranking showed moderate clinical priority for all antidepressants, with the exception four, that had a weak one (figure 1). Most frequently reported symptoms were respiratory symptoms (n=106), irritability/agitation (n=75), tremor (n=52) and feeding problems (n=40).
Image:
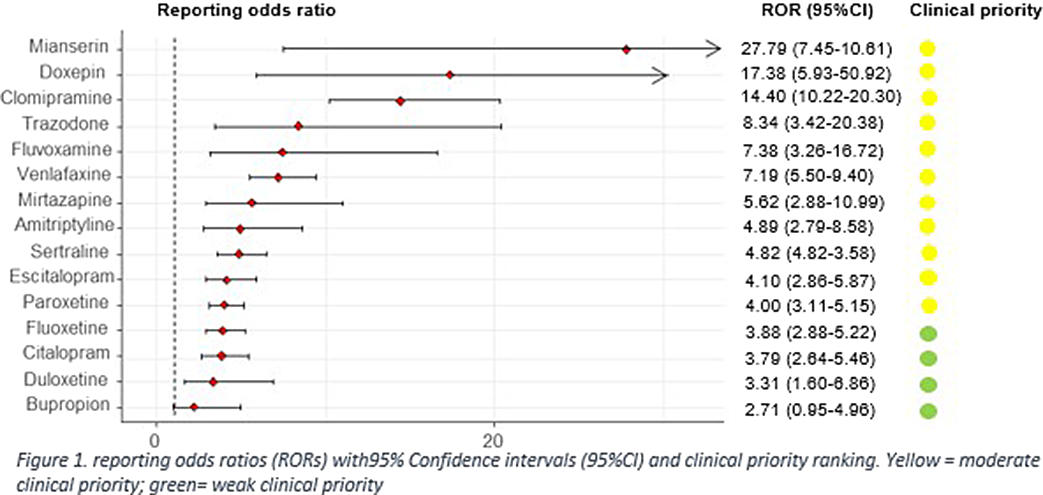 Conclusions
ConclusionsExposure to antidepressants in utero is associated with moderate signals of disproportionate reporting for neonatal withdrawal syndrome for most antidepressants. Clinicians should pay extra attention to neonates with antidepressant-treated mothers.
Disclosure of InterestC. Gastaldon: None Declared, E. Arzenton : None Declared, E. Raschi: None Declared, O. Spigset: None Declared, D. Papola: None Declared, G. Ostuzzi: None Declared, U. Moretti: None Declared, G. Trifirò Grant / Research support from: he was the scientific director of a Master program on pharmacovigilance, pharmacoepidemiology and real-world evidence which has received non-conditional grant from various pharmaceutical companies; he coordinated a pharmacoepidemiology team at the University of Messina until Oct 2020, which has received funding for conducting observational studies from various pharmaceutical companies (Boehringer Ingelheim, Daichii Sankyo, PTC Pharmaceuticals). He is also scientific coordinator of the academic spin-off “INSPIRE srl” which has received funding for conducting observational studies from contract research organizations (RTI Health Solutions, Pharmo Institute N.V.)., Consultant of: has served in the last three years on advisory boards/seminars funded by SANOFI, Eli Lilly, AstraZeneca, Abbvie, Servier, Mylan, Gilead, Amgen, Speakers bureau of: has served in the last three years on advisory boards/seminars funded by SANOFI, Eli Lilly, AstraZeneca, Abbvie, Servier, Mylan, Gilead, Amgen, C. Barbui: None Declared, G. Schoretsanitis Consultant of: Dr. Schoretsanitis has served as a consultant for HLS Therapeutics and Thermo Fisher Scientific.
Neonatal withdrawal syndrome following in utero exposure to antidepressants: a disproportionality analysis of VigiBase, the WHO spontaneous reporting database
- C. Gastaldon, E. Arzenton, E. Raschi, O. Spigset, D. Papola, G. Ostuzzi, U. Moretti, G. Trifirò, C. Barbui, G. Schoretsanitis
-
- Journal:
- Psychological Medicine / Volume 53 / Issue 12 / September 2023
- Published online by Cambridge University Press:
- 21 September 2022, pp. 5645-5653
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Background
Evidence on neonatal withdrawal syndrome following antidepressant intrauterine exposure is limited, particularly for antidepressants other than selective serotonin reuptake inhibitor (SSRIs).
MethodsIn our case/non-case pharmacovigilance study, based on VigiBase®, the WHO database of suspected adverse drug reactions, we estimated reporting odds ratio (ROR) and the Bayesian information component (IC) with 95% confidence/credibility intervals (CI) as measures of disproportionate reporting of antidepressant-related neonatal withdrawal syndrome. Antidepressants were first compared to all other medications, then to methadone, and finally within each class of antidepressants: SSRIs, tricyclics (TCA) and other antidepressants. Antidepressants were ranked in terms of clinical priority, based on semiquantitative score ratings. Serious v. non-serious reports were compared.
ResultsA total of 406 reports of neonatal withdrawal syndrome in 379 neonates related to 15 antidepressants were included. Disproportionate reporting was detected for antidepressants as a group as compared to all other drugs (ROR: 6.18, 95% CI 5.45–7.01, IC: 2.07, 95% CI 1.92–2.21). Signals were found for TCAs (10.55, 95% CI 8.02–13.88), followed by other antidepressants (ROR: 5.90, 95% CI 4.74–7.36) and SSRIs (ROR: 4.68, 95% CI 4.04–5.42). Significant disproportionality emerged for all individual antidepressants except for bupropion, whereas no disproportionality for any antidepressant was detected v. methadone. Eleven antidepressants had a moderate clinical priority score and four had a weak one. Most frequent symptoms included respiratory symptoms (n = 106), irritability/agitation (n = 75), tremor (n = 52) and feeding problems (n = 40).
ConclusionsMost antidepressants are associated with moderate signals of disproportionate reporting for neonatal withdrawal syndrome, which should be considered when prescribing an antidepressant during pregnancy, irrespective of class.





